aScope™ Gastro and aScope™ Gastro Large
The Ideal Disposable Gastroscopes for Critical Care and Surgical Endoscopy
Single-Use Enables Hassle-Free Endoscopy From the ICU to the Operating Room
The new Ambu® aScope™ Gastro Large is the first gastroscope with a 4.2 mm working channel. We designed it to pair with the Ambu® aScope™ Gastro to offer a completely portable, GI portfolio.
Together, these single-use solutions:
- Can tackle a wider array of EGD indications
- Enable you to bring the necessary scope where it’s needed — be it the ICU, OR, or endoscopy suite
- Are available around the clock—whenever, wherever

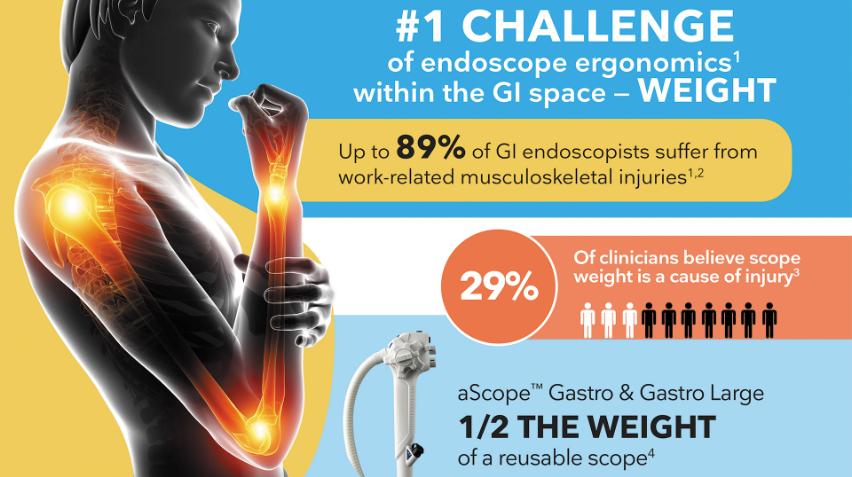
Ergonomics | Single-Use Scopes Half the Weight of Reusables
If heavy endoscopes add to your fatigue, consider single-use scopes. Studies show:
Up to 89% of GI endoscopists suffer from musculoskeletal injuries (Tech. GI Endoscopy)
29% of clinicians believe scope weight is a cause of injury (Endoscopy Int Open)
Scopes in our aScope Gastro portfolio are half the weight of reusables, which means less fatigue and more endurance for lengthy procedures.
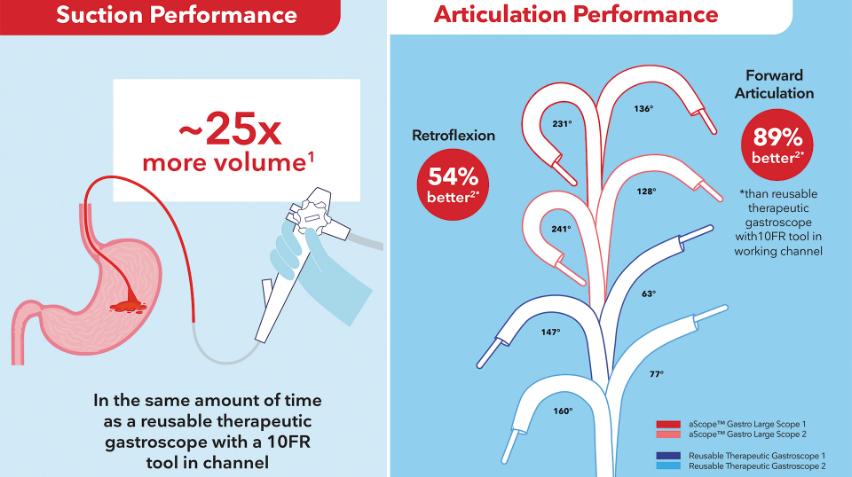
New Research | Better Suction, Articulation Performance
Two new studies detailed how the Ambu aScope Gastro Large delivered superior performance over reusables:
- Aspirates approximately 25x more volume in same time as the leading reusable therapeutic gastroscope
- Superior articulation performance in both directions (54% better retroflexion)
In critical cases like acute upper GI bleeding, powerful suction can mean everything.
The GI Community on the aScope Gastro
ACCESSIBILITY | General surgeon Dr. Jawad Ali discusses how the aScope Gastro's accessiblity can make the difference in delivering faster patient care.
RETROFLECTION | Dr. Aram Jawed uses the aScope Gastro to determine the presence of a hiatal hernia following a sleeve gastrectomy.
BEDSIDE EVALUATIONS | Dr. Vani Konda discusses the benefits of using the aScope Gastro for GI bedside evaluations.
WORKFLOW BOOST | Bariatric surgeon Dr. Morris Washington explains how the Ambu aScope Gastro boosted his facility's workflow.
SEE WHAT YOU CAN DO WITH 4.2 - Superior Suction With the aScope Gastro Large
The aScope Gastro Large, with a 4.2 mm working channel, offers 90% improved suction performance vs. the industry-leading reusable therapeutic gastroscope
Four Procedures Where a Large Gastroscope Working Channel May Offer Clinical Advantages
Acute Upper Gastrointestinal Bleeding (UGIB)
One of the most common gastrointestinal emergencies for which flexible endoscopy has become an indispensable tool.
Advantages of a large working channel:
•Provides increased aspiration
•Efficient removal of blood, clots and debris
•Allows use of a broad range of therapeutic instruments for efficient hemostasis
Direct Endoscopic Necrosectomy (DEN)
Can be employed, along with endoscopic drainage, to treat walled-off pancreatic necrosis (WOPN) after acute pancreatitis.
Advantages of a large working channel:
•Facilitates increased aspiration for removal of necrotic material
•Allows for use of large-caliber instruments for debridement and TTS stents
Stricture Management
Involves endoscopic dilation and stenting to treat upper gastrointestinal luminal obstruction.
Advantages of a large working channel:
•Enables direct visualization and deployment of through-the-scope stents
•May help minimize reliance on fluoroscopy
Removal Of Foreign Bodies And Food Impaction
Endoscopy plays an essential role in the management of foreign body ingestion and food impaction.
Advantages of a large working channel
•Allows for use of a wide variety of retrieval tools
•Provides increased aspiration power for application of endoscopic suction technique

Excellent Imaging, Intuitive Design
Get a live endoscopic HD image on the built-in touchscreen within seconds when you connect aScope Gastro to the aBox 2. The intuitive user interface is right on the screen next to the endoscopic image, making it easy to maintain focus and control. The touchscreen can be used as either the primary or the secondary monitor, and it adapts perfectly to different needs and room setups.
- Full-HD LCD monitor for excellent imaging
- Advanced image processing, adaptive light control, and true colors
- Take images and record videos at the touch of a button on aScope Gastro or aBox 2
- Easy transfer of recorded imaging data to PACS using DICOM over Wi-Fi or LAN
- Digital video output options to display live image on external monitors
Lowering Our Carbon Footprint
The aScope Gastro Large is the first endoscope made with bioplastics, elevating sustainability standards. By 2025, we will use bioplastics in every endoscope handle we produce.
Are You Ready to Unlock EGD Efficiency with Our aScope Gastro Platform?
See our product in action! Fill out the contact form below to request additional info or to receive a customized, one-on-one product demo at your convenience from a local Ambu representative. Discover how our aScope Gastro scopes can deliver reliable performance for you.
Evidence
The aScope Gastro has already been featured in many clinical and efficiency studies

