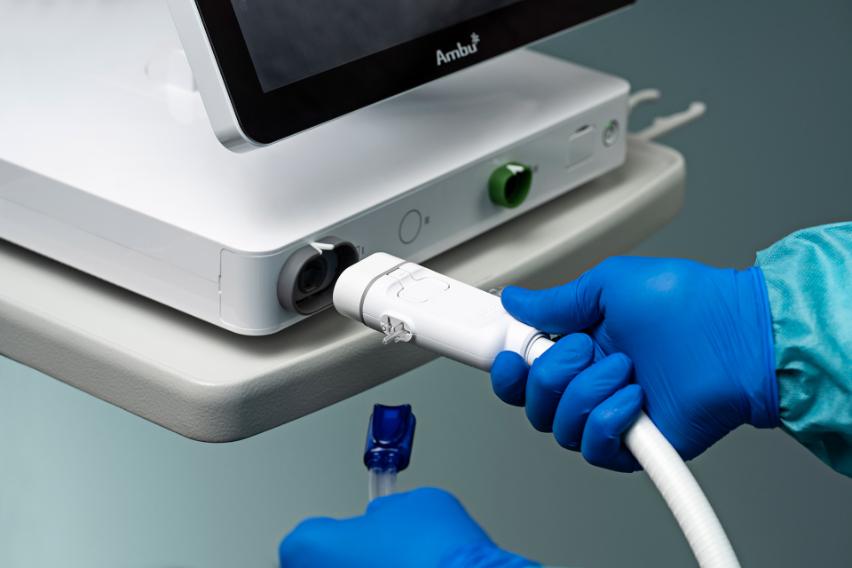
aScope™ Duodeno 2
Ready to Start ERCP — But Your Scope Isn’t?
Nights and Weekends When Resources Are Limited – No Reprocessing Required
ERCP procedures don’t always go according to plan. When endoscopes are still in reprocessing, being transported, or unavailable, procedures are delayed and schedules are disrupted.
The Ambu® aScope™ Duodeno 2 allows teams to proceed with ERCP procedures without waiting for reprocessing or turnover. Elevate your workflow, don’t interrupt It.
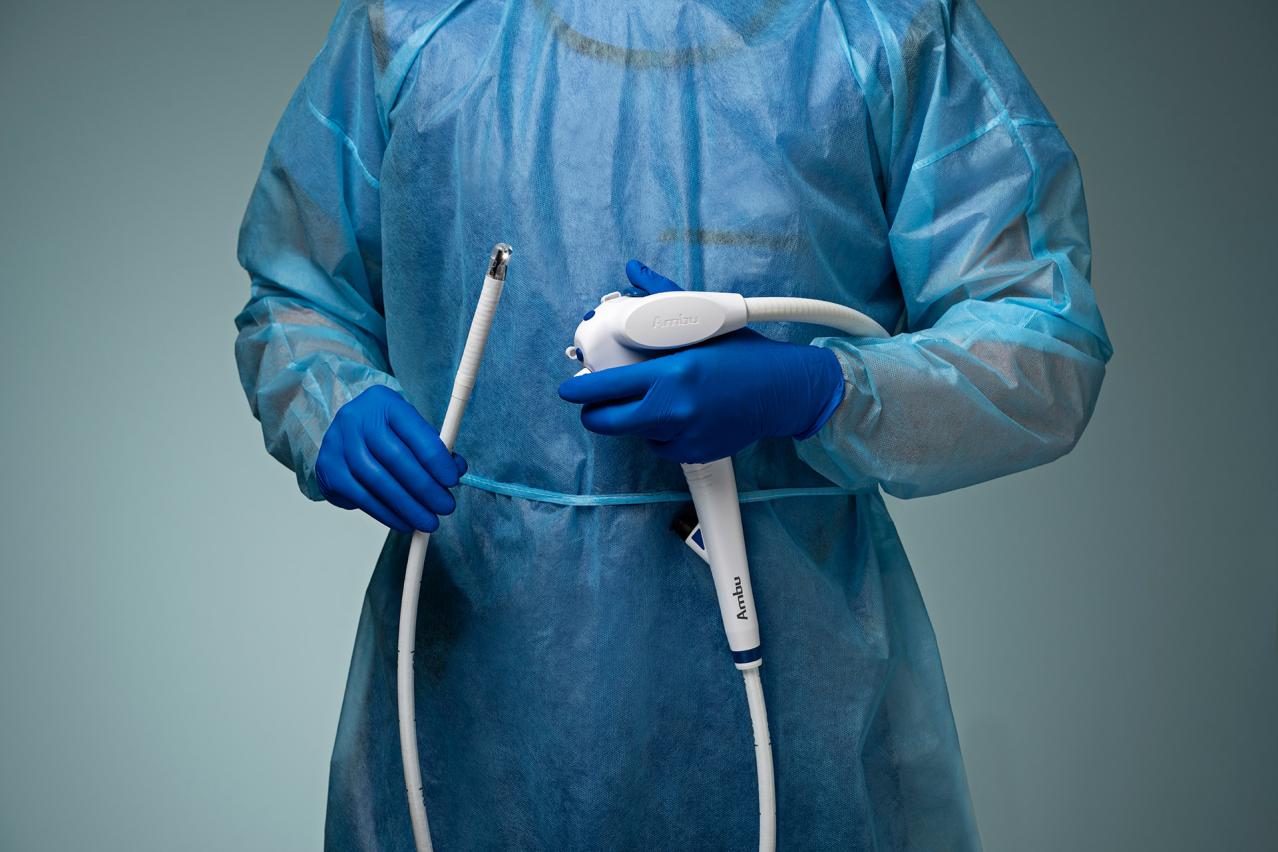
See How aScope Duodeno 2 Can Elevate Your Workflow - Not Disrupt It.
Discover how aScope Duodeno 2 enables you to maintain your ERCP workflow efficiency even on your busiest days, after-hours, and weekends. Complete the contact form below to request additional information or to schedule a demonstration of the aScope Duodeno 2 at your convenience with your local Ambu representative.
When the Scope Isn’t Ready, the Entire Team Feels It
When a duodenoscope is unavailable or still in reprocessing, the entire ERCP schedule can slow down – impacting workflow across the entire team.
The aScope Duodeno 2 is always available and arrives sterile, fully assembled with valves installed, and ready to use, allowing teams to proceed without waiting for endoscope preparation.
Learn more about Ambu single-use endoscopy solutions supporting workflow across GI procedures
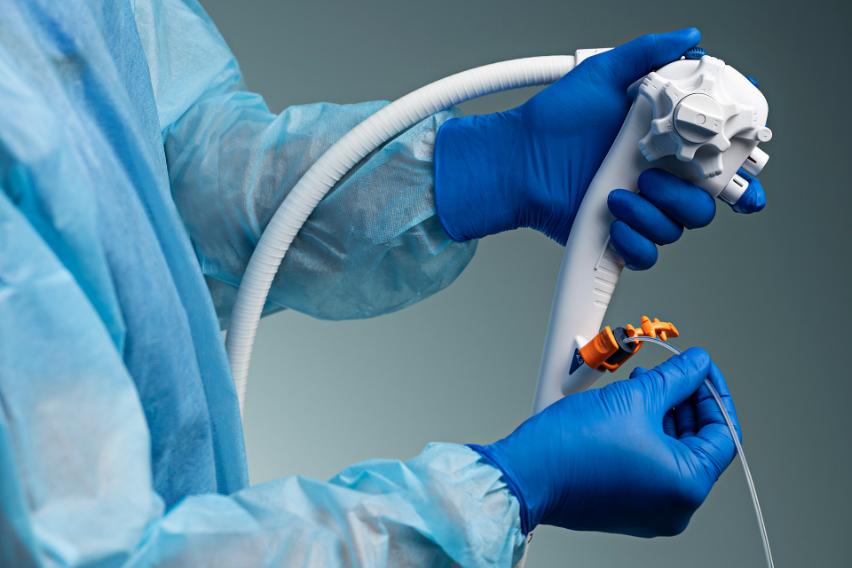
A Sterile Option for More Vulnerable Patients
For patients with known infections or elevated clinical risk, the aScope Duodeno 2 is sterile and straight out of the package.
The aScope Duodeno 2 is the first duodenoscope with bioplastic material in the handle, supporting Ambu’s sustainability initiatives and circular economy programs.
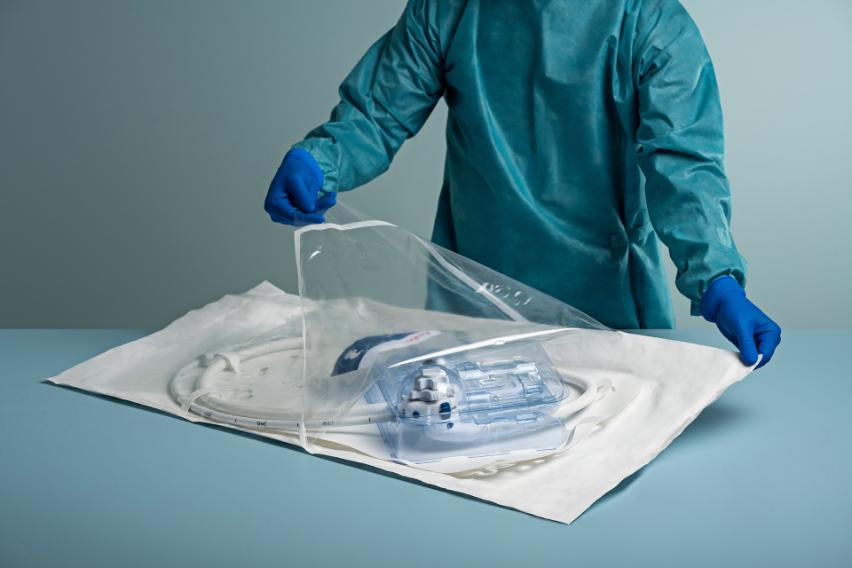
Built for ERCP – Enhance Performance with Innovative Features
- Ergonomic Handle – Supports a neutral wrist position during procedures
- Radiolucent Distal End – Improves visualization during fluoroscopy
- Integrated Valves – Arrives fully assembled, ready to use with valves installed
- Full HD Imaging – Compatible with the Ambu® aBox™ 2 Endoscopy System
Learn more about the Ambu® aBox™ 2 Endoscopy System